The U.S. Food and Drug Administration (FDA) doesn’t just check boxes during inspections. It’s looking for systemic failures that could put patients at risk. In 2025, warning letters issued to pharmaceutical and medical device manufacturers revealed a clear pattern: the biggest threats aren’t always high-tech problems. They’re simple, preventable breakdowns in how companies manage quality. These aren’t isolated mistakes. They’re signs of deeper issues - and they’re costing companies access to the U.S. market.
Aseptic Processing: The Silent Killer
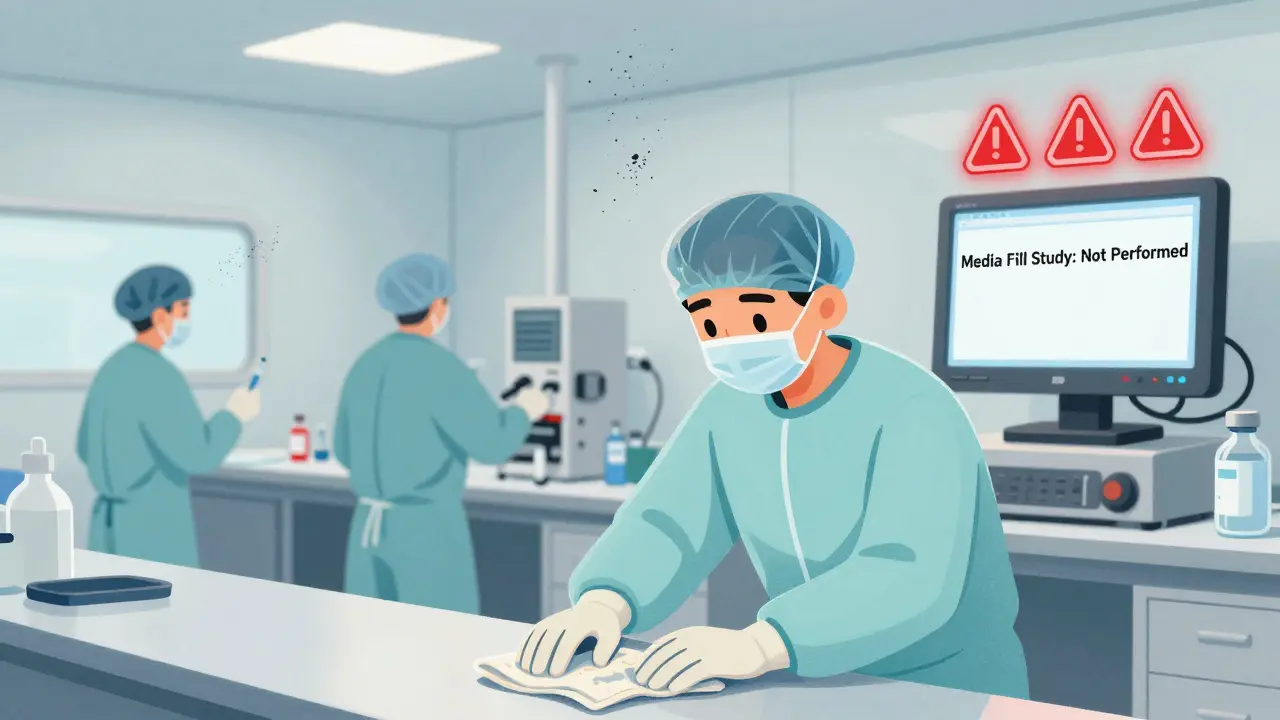
If you’re making injectable drugs or sterile medical devices, a single contaminated batch can kill. That’s why the FDA zeroes in on aseptic processing - and it’s the number one issue in 2025. Nearly half of all warning letters (47%) cited failures here. What does that look like in real life?
- A facility skips media fill studies - the mock runs that test whether sterile conditions hold up during actual production.
- Workers don’t change gloves or gowns properly between steps, dragging contaminants into clean zones.
- Airflow in cleanrooms is inconsistent, and no one’s monitoring it in real time.
The FDA doesn’t accept excuses like "we’ve never had a problem." One company, Health and Natural Beauty USA Corp., got flagged in July 2025 for failing to prove their sterile filling process worked. They were told: "No data means no safety." The result? Their products were blocked from entering the U.S. until they fixed it.
Data Integrity: When Records Lie
It’s not just about having records. It’s about having records you can trust. The FDA calls this ALCOA+: data that’s Attributable, Legible, Contemporaneous, Original, Accurate, and complete. In 2025, 39% of warning letters mentioned data integrity failures. This isn’t about sloppy handwriting anymore. It’s digital.
- A lab uses UV-Vis instruments without audit trails. Someone changes a result - and there’s no record of who did it or when.
- Production teams write batch records on laminated paper and erase entries with erasable markers. The FDA saw this at a facility in China.
- Electronic logs are turned off overnight to "avoid clutter." That’s a violation.
The FDA doesn’t just want clean records. They want proof that no one tampered with them. One warning letter to Guangxi Yulin Pharmaceutical Group specifically called out missing audit trails on their infrared spectrometers. Without those trails, there’s no way to know if a test result was accurate - or altered.
Material Control: Bad Ingredients, Bad Outcomes
Garbage in, garbage out. That’s the FDA’s mindset when it comes to raw materials. In 35% of 2025 warning letters, companies failed to properly test or verify what they were putting into their products. The most alarming example? Glycerin and sorbitol contaminated with diethylene glycol (DEG) - a toxic substance linked to kidney failure and death.
- A company in Malaysia didn’t test incoming glycerin for DEG, even though their supplier’s certificate of analysis was outdated.
- Another firm trusted a supplier’s test results without doing their own verification. The FDA found this at Foshan Yiying Hygiene Products Co., Ltd.
The FDA expects manufacturers to go beyond paper certificates. They need to test high-risk materials themselves, using methods that can detect contaminants at levels as low as 0.1% w/w. USP General Chapter <1085> spells this out clearly. Ignoring it isn’t negligence - it’s a public health risk.

Process Validation: Guessing Isn’t a Strategy
Just because a process worked once doesn’t mean it’s reliable. The FDA requires proof - not just for drugs, but for toothpaste, hand sanitizers, and medical device components too. In 28% of 2025 warning letters, companies couldn’t show they validated their manufacturing methods.
- A company making toothpaste had no validation study for their mixing process. How did they know the active ingredient was evenly distributed?
- Another firm used a method to test purity, but never proved the method actually worked. The FDA called this "scientifically unsound."
Validation isn’t a one-time task. It’s ongoing. The FDA requires three consecutive batches to meet all specifications before a process is considered validated. And every step - from raw material handling to final packaging - needs documented controls.
The Hidden Problem: Quality Culture
Beneath every technical failure is a cultural one. Dr. David Lim from Compliance Architects says 78% of companies cited in 2025 warning letters had a leadership culture that valued speed over safety. That’s the real root cause.
- Managers pressure teams to skip tests to meet deadlines.
- Quality units are underfunded and have no authority to stop production.
- Employees are afraid to report problems because they’ll be blamed, not supported.
The FDA saw this clearly in a warning letter to a Chinese facility: "This site does not prepare batch production records for every batch of our listed drug." That’s not a mistake. That’s a culture. The FDA’s Quality Management Maturity (QMM) initiative was created to spot this early. Facilities that fix their culture see 63% fewer repeat issues.

Where It’s Happening - And Why
It’s not random. In 2025, 73% of warning letters targeted manufacturers in China, India, and Malaysia. Each region has its own pattern:
- China: 28 warning letters cited failures in analytical method validation. Many labs use outdated methods or don’t prove they’re accurate.
- India: 24 letters focused on data integrity. The CDSCO, India’s drug regulator, inspects fewer than 2% of domestic facilities each year - leaving gaps the FDA has to fill.
- Malaysia: 9 letters pointed to weak quality units. Quality teams lacked authority to halt production, even when problems were found.
The FDA responded by increasing unannounced inspections by 40% in 2025, with 68% of those targeting Asian facilities. They’re not guessing anymore. They’re following the data.
What Happens When You Get Caught
Getting a warning letter isn’t the end - but it’s the beginning of a long, expensive fix. Here’s what the FDA typically demands:
- Independent consultant: 92% of 2025 letters required hiring an outside CGMP expert to review and fix systems.
- Data system overhaul: Electronic records must have audit trails, user access controls, and 180-day retention. No exceptions.
- Re-validation: For manufacturing processes, you must produce three successful batches under strict controls.
- Import Alert: 147 facilities were placed on Import Alert 66-60. Their products are now stopped at U.S. ports until they prove they’re safe.
Remediation takes 6 to 18 months. And it’s not cheap. Global spending on CGMP compliance hit $4.7 billion in Q3 2025 - up 12.3% from the year before.
What’s Next in 2026
The FDA isn’t slowing down. In fact, they’re doubling down:
- Unannounced inspections will jump from 850 in 2025 to 1,200 in 2026 - including U.S. facilities for the first time.
- QMM assessment results will start influencing inspection frequency. Companies with strong quality cultures may get fewer visits.
- New focus areas: cloud-based quality systems, oversight of contract labs, and continuous manufacturing processes.
One thing is clear: the FDA is no longer just checking for compliance. They’re checking for culture. If your team sees quality as a cost center, not a core value - you’re already behind.
What are the most common FDA manufacturing deficiencies in 2025?
The top three deficiencies in 2025 were aseptic processing failures (47% of warning letters), data integrity issues (39%), and material control lapses (35%). These reflect systemic breakdowns in sterile production, record-keeping, and raw material verification - not isolated mistakes.
Can a company fix an FDA warning letter without hiring a consultant?
Technically, yes - but practically, no. In 2025, 92% of warning letters required hiring an independent CGMP consultant. The FDA doesn’t trust internal teams to fix their own failures. An outside expert brings objectivity, experience, and credibility needed to satisfy regulators.
Why does the FDA focus so much on data integrity?
Because if you can’t trust your data, you can’t trust your product. Data integrity failures mean test results might be fake, batch records could be altered, and safety claims aren’t backed by evidence. The FDA’s ALCOA+ standards ensure records are accurate, traceable, and tamper-proof - essential for patient safety.
What’s the difference between a 483 observation and a warning letter?
A Form 483 is a list of inspection findings given at the end of a visit. It’s a heads-up. A warning letter is a formal legal notice that the FDA believes you’ve violated federal law. It comes after you’ve had time to respond to the 483 - and if the FDA isn’t satisfied, they escalate. Warning letters can lead to import bans or lawsuits.
How can a manufacturer avoid FDA enforcement actions?
Build a strong quality culture. Train staff to speak up without fear. Empower the quality unit to stop production. Validate every process. Test materials rigorously. Maintain full audit trails. And don’t cut corners to meet deadlines. The FDA rewards companies that treat quality as a priority - not a hurdle.
Deborah Dennis
March 3, 2026 AT 17:51Diane Croft
March 4, 2026 AT 10:09Donna Zurick
March 6, 2026 AT 00:55Justin Rodriguez
March 8, 2026 AT 00:32tatiana verdesoto
March 9, 2026 AT 02:56Ethan Zeeb
March 11, 2026 AT 01:43Megan Nayak
March 12, 2026 AT 17:17marjorie arsenault
March 13, 2026 AT 05:46Darren Torpey
March 15, 2026 AT 02:53Matt Alexander
March 16, 2026 AT 02:22Levi Viloria
March 16, 2026 AT 03:42Sharon Lammas
March 17, 2026 AT 15:52Mariah Carle
March 18, 2026 AT 04:43