Category: Pharmacy Guide - Page 2
- April 18 2026
- 0 Comments
- Colin Winthrop
How to Buy Cheap Generic Zovirax Online: Pricing & Pharmacy Guide
Learn how to save money on generic Zovirax (acyclovir) with this guide to online pharmacies, prescription coupons, and price-matching strategies for 2026.
- April 10 2026
- 0 Comments
- Colin Winthrop
Beyond the Label: Reshaping How We View Generic Drugs
Explore the brand psychology behind generic drugs and the future strategies-from biosimilars to AI transparency-used to bridge the perception gap between cost and quality.
- April 7 2026
- 10 Comments
- Colin Winthrop
MAOIs and OTC Cold Medicines: Avoiding Hypertensive Crisis and Serotonin Syndrome
Learn why mixing MAOIs with OTC cold medicines can cause hypertensive crisis and serotonin syndrome. Discover safe alternatives and critical warning signs.
- March 29 2026
- 10 Comments
- Colin Winthrop
Statin Interactions with Other Medications: Myopathy Risks Explained
Discover how statin drug interactions trigger myopathy and rhabdomyolysis risks. Learn which antibiotics, heart meds, and supplements cause dangerous spikes, plus safe alternatives.
- March 27 2026
- 10 Comments
- Colin Winthrop
The Hidden Dangers of Skipping Prescribed Medications
Skipping prescriptions kills 125,000 Americans yearly. Learn the hidden costs, health risks, and practical steps to manage medication adherence safely.
- March 24 2026
- 9 Comments
- Colin Winthrop
Metronidazole and Alcohol: What Really Happens When You Mix Them
New research shows metronidazole doesn't cause a dangerous reaction with alcohol - contrary to decades of medical advice. Learn what really happens and what you should do.
- March 21 2026
- 13 Comments
- Colin Winthrop
MedWatch: How to Report Medication Problems and Side Effects
Learn how to report medication side effects and adverse reactions through MedWatch, the FDA's official system for tracking drug safety. Your report can help prevent harm to others.
- March 7 2026
- 14 Comments
- Colin Winthrop
Pharmacy Verification Services: How to Check Online Pharmacy Licenses
Learn how to verify online pharmacy licenses to avoid counterfeit drugs and unsafe vendors. Discover how state systems and NABP Verify work, what to look for, and how to protect yourself.
- March 6 2026
- 8 Comments
- Colin Winthrop
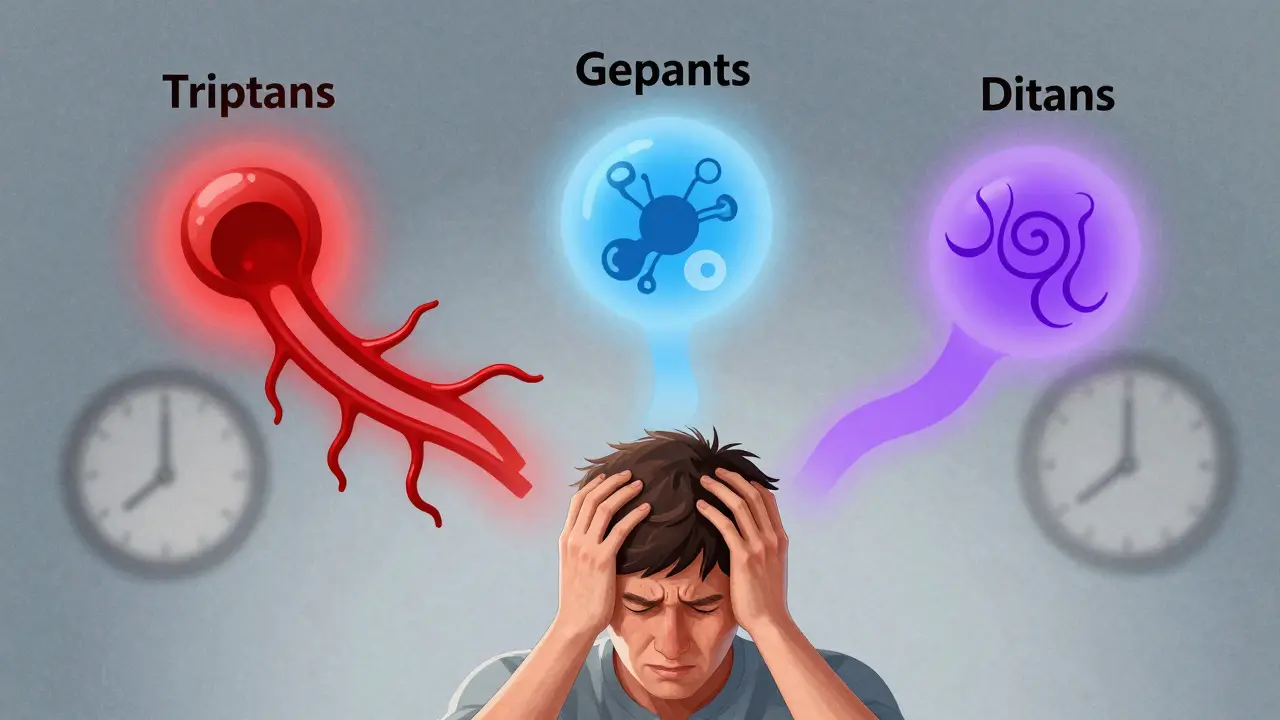
Migraine Medications: Triptans, Gepants, and Ditans Safety Compared
Triptans, gepants, and ditans offer different migraine relief options with varying safety profiles. Learn which is safest for heart patients, drivers, and daily users based on clinical data and real-world feedback.
- March 3 2026
- 13 Comments
- Colin Winthrop
Manufacturing Deficiencies: Common Quality Issues Found by FDA in 2025
In 2025, the FDA issued warning letters exposing widespread manufacturing flaws - from contaminated ingredients to falsified records. These aren't technical glitches. They're signs of failing quality cultures that put patients at risk.
- February 21 2026
- 9 Comments
- Colin Winthrop
Timing Medication Doses: How Administration Time Affects Side Effects
The time you take your medication can significantly impact its effectiveness and side effects. From statins to insulin, circadian rhythms influence how drugs work in your body. Learn which meds need strict timing and how to get it right.
- February 17 2026
- 9 Comments
- Colin Winthrop
Antibiotic-Related Liver Injury: Hepatitis and Cholestasis Explained
Antibiotics can cause liver injury in the form of hepatitis or cholestasis, with amoxicillin-clavulanate being the most common culprit. Learn how it happens, who’s at risk, and what doctors do to prevent and manage it.
- Pharmacy Guide (128)
- Health & Wellness (64)
- Health and Wellness (1)
- Water Treatment (1)
- Legal Updates (1)
Categories
- June 2026 (1)
- May 2026 (14)
- April 2026 (8)
- March 2026 (11)
- February 2026 (12)
- January 2026 (29)
- December 2025 (29)
- November 2025 (19)
- October 2025 (30)
- September 2025 (14)
- August 2025 (7)
- July 2025 (26)
Archives
- online pharmacy
- generic drugs
- side effects
- drug interactions
- drug safety
- pharmacy guide
- antibiotics
- safe online pharmacy
- sleep apnea
- statin side effects
- online pharmacy safety
- ED medication
- prescription discount
- acne treatment
- antibiotic
- medication interactions
- hyaluronic acid supplement
- skin hydration
- Sildenafil
- alternatives